Log In
 Ralph & Bev Shock
Ralph & Bev Shock 
Chemical Formulas
As we saw in chapter 2 a chemical name for an element can be abbreviated with a chemical symbol. Usually the symbol contains the first letter of the element's name. If two symbols begin with the first letter the two letters will be used and the second letter is always lowercase. Example: sulfur is S and silicon is Si. Be careful to always write the second letter as lowercase because confusion can occur with elements and compounds.Example: Co is cobalt and CO is the molecular compound carbon monoxide.
For writing chemical formulas it is important to make oneself familiar with the periodic table. The elements are arranged in rows and columns. These arrangements correspond to the chemical properties of the elements.
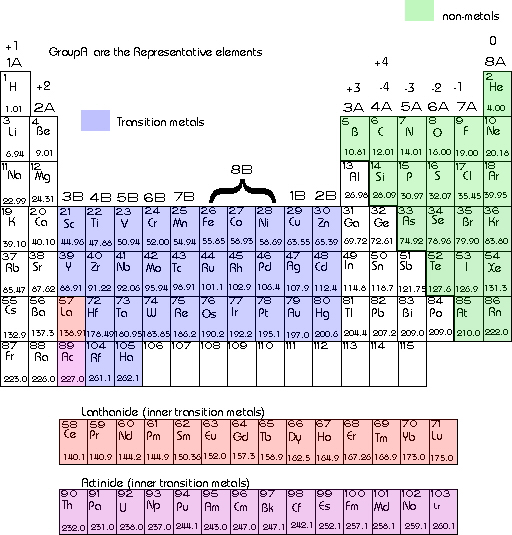
A column on the periodic table is known as a group. Each group is designated by a number-letter combination. The Group 1A elements contain only one electron in their outer energy level. They will typically lose one electron to obtain an electron configuration of the preceeding noble gas. This means that all the elements in group 1A have charges of +1 when forming ionic compounds. All the elements in group 2A have two electrons in their outer energy level and correspondingly obtain charges of +2. For all of the group A elements the group number corresponds to the number of electrons in the outer energy level. Since these electrons are involved in bonding they give an indication of how many electrons will be lost, gained or shared during bonding. Nonmetals typically gain electrons when forming ionic bonds so they will take on negative charges. For example Chlorine has 7 electrons in its outer energy level and therefore will gain one electron developing a -1 charge.
A chemical formula is an easy and clear way to represent compounds and the elements that they are comprised of. The chemical formula also gives you the ratios of the atoms in a chemical compound. Thus the chemical formula H2O gives you the kinds and numbers of atoms bonded together in water.
Compounds that consist of only two different elements are called binary compounds. Examples include sodium chloride, dinitrogen monoxide, silicon dioxide, calcium fluoride. There formulas are NaCl, N2O, SiO2, CaF2 respectively. Notice that their names all end in ide, the names of all binary compounds end in ide.
There are two major types of chemical compounds based on the chemical bonds holding the atoms or groups of atoms together.
1. Ionic bonds are formed when atoms transfer electrons to achieve electron configurations like noble gases (8 electrons in their outer sublevels (Typically s & p sublevels)). The negative ion is called an anion and the positive ion is called a cation.
2. Molecular bonds are formed when atoms share electrons to achieve electron configurations like noble gases (8 electrons in their outer sublevel).
Ionic compounds can be recognized by the fact that they are usually made up of a metal and nonmetal in binary compounds or contain polyatomic ions in tertiary compounds.
You must be able to correctly determine chemical formulas based on the charges of the ions.
Remember all compounds are electrically neutral.
Example: The compound made from Lithium (Li )and Sulfur (S) must be Li2S because lithium forms a charge of +1 when it loses one electron and sulfur forms a charge of -2 when it gains two electrons, therefore two lithium atoms are needed to balance the -2 charge of the one sulfur atom.
2Li+1 + S-2 ---------> Li2S (2x1)+(1x(-2))=0 The charge of a ionic compound must always be zero!
Notice that each element now has an electron configuration of a Nobel gas; lithium -1 that of helium (2 electrons) and sulfur 2- has the same number of electrons as argon (2,8 and 8 electrons ). These new electron configurations make them more stable than they would have in their elemental form. A bond is formed because of the opposite charges of the ions. These are called electrostatic attractions, like static electricity.
The same procedure applies to tertiary compounds containing polyatomic ions. Polyatomic ions are groups of covalently bonded atoms (molecular bonds) with an overall charge. The whole group it to be treated as a single ion. Polyatomic ions are to be treated as a single charged group. If there is more than one polyatomic ion needed to balance the charge then the group must be put in parenthesis and the subscript written to the outside.
Example: Fe(ClO)3 is the formula for Iron (III) hypochlorite . The Roman numeral tells us that iron has a charge of +3, and the polyatomic ion hypochlorite has a charge of -1, therefore we need three hypochlorite ions bonded to the one iron atom.
Example: Al2(SO4)3 for aluminum sulfate. Aluminum always has a charge of +3 and the sulfate polyatomic ion (SO4-2) has a charge of -2. In order to balance the charge and make it zero it will take two aluminum ions of charge +3 and three sulfate ions of -2 to balance the charge.
2Al+3 + 3SO4-2 --------> Al2(SO4)3 (2x3)+(3x(-2))=0 Notice that the charges are crisscrossed to the absolute value of the subscript. Al2+3(SO4-2)3 This is always true for ions with even and odd charges but note that a subscript is not used for one ion like in Fe(ClO)3 the one atom of iron is understood without the subscript.
When naming ionic compounds the names of just the ions are used: All binary compounds end in -ide. This is because the monoatomic anion ends in ide.
Example Ca3P2 is Calcium Phosphide. The two ions are the Calcium ion Ca+2 and the Phosphide mono atomic anion is P-3. Note the ending of all elemental anions is always ide. If the compound contains a transition metal or metal underneath the stair-step that separates the metals from the nonmetals then you must use a Roman numeral. There are four exceptions to this rule: Al, Cd, Ag, Zn. This is because they only ever have one charge Al3+, Cd2+, Ag1+, Zn2+.
Example: Fe3P2 is Iron(II) Phosphide.
Example: V2S5 is Vanadium (V) Sulfide. Note that the charge can be determined on Vanadium because sulfur has a charge of -2 and there are 5 of them. (5x-2)=-10. Since there are 2 Vanadiums its charge has to be +5.
Example: CdSe is just Cadmium Selenide because even though Cadmium is a Group B element (Transition Metal) it is an exception because it can only have one charge (+2).
For ionic compounds containing polyatomic ions you just name the ions:
Example: Ca3PO4 is Calcium phosphate. All but three of the polyatomic ions that you have learned either end in ate or ite. The exceptions are hydroxide, cyanide and ammonium.
If a tertiary compound contains a transition metal or metal underneath the stair-step that separates the metals from the nonmetals then you must use a Roman numeral.
Example: FeHPO4 is Iron (II) hydrogenphosphate.
Example: (NH4)2C2O4 is ammonium oxalate . The two ions are the ammonium ion and the oxalate ion.
There are four exceptions to this Zn, Cd, Ag, & Al. With these four metals you do not use a Roman numeral.
Example: Ag2SiO3 is Silver Silicate (no Roman numeral is necessary)
Molecular compounds can be recognized by the fact that they usually only contain nonmetals.
When naming molecular compounds you must use prefixes to represent the number of each kind of atom. Prefixes are:
mono - 1, di - 2, tri - 3, tetra - 4, penta - 5, hexa - 6, hepta - 7, octa - 8, nona - 9, deca - 10.
Note: Do Not Worry About Charges in Molecular Compounds. They are sharing electrons!
Example: P2O5 is diphosphorus pentoxide. If there is only one atom of the first element you don't use the prefix mono.
Example: CO2 is carbon dioxide.
If there is only one atom of the second element then you must use the prefix mono.
Example: CO is carbon monoxide.
Example: SCl6 is sulfur hexaflouride.
Example: P4S10 is tetraphosphorous decasulfide.
Notice the first element is just the name of the element and the second element ends in ide.
Certain elements only exist in a binary molecule made up of two identical atoms covalently bonded together.
They are hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine. When these elements are not in compounds; that is when they are by themselves they are never to be written as a single elemental symbol in chemical equations or in their elemental form. They are always written as:
H2, N2, O2, F2, Cl2, Br2, I2
Acids are molecular compounds that ionize in water releasing hydrogen ions.
Example: HCl is the molecular compound hydrogen monochloride but when it is dissolved in water the polarity of water causes it to break apart (dissociate) into a H+1 ion and a Cl-1 ion. The release of hydrogen makes it an acid.
Acids can be recognized by the fact that the first element in the compound is hydrogen:
Example: HCl. Binary acids like HCl all begin with hydro. The ide of there nonacid name is changed to ic and acid is added at the end.
Therefore HCl is hydrochloric acid. Other examples are H2S (hydrosulfuric acid), HBr (hydrobromic acid), H2Se (hydroselenic acid). The real overriding rule is that all acids in which the "anion" ends in ide the acid begins with hydro the ide ending is changed to ic and acid is added.
Since the polyatomic ion cyanide ends in ide it is the only tertiary acid that begins with hydro. Therefore its name is hydrocyanic acid.
All other tertiary acids do not begin with hydro.
If the non acid anion ending of a polyatomic ion is ate, the ate is changed to ic and acid is added.
Example: H2SO4 is sulfuric acid.
If the non acid anion ending of a polyatomic ion is ite, the ite is changed to ous and acid is added.
Example H2SO3 is sulfurous acid.
The following flowchart is very useful for naming compounds.

Example Test
MATCHING:
| Column A | Column B |
| 1. law of definite proportions | a. shows the number and kinds of atoms present in a molecule of a compound. |
| 2. molecular formula | b. tightly bound groups of atoms that behave as a unit and carry a charge |
| 3. cation | c. when two elements form more that one compound, the masses of one element are in the ratio of small whole numbers. |
| 4. ternary compound | d. a compound composed of two different elements |
| 5. chemical formula | e. Group B elements |
| 6. metalloid | f. any atom or group of atoms with a positive charge |
| 7. law of multiple proportion | g. a compound composed of three different elements |
| 8. transition metal | h. in any chemical compound, the elements are combined in the same proportion by mass |
| 9. binary compounds | i. elements with the properties of both metals and nonmetals |
| 10. Polyatomic ion. | j. shows the kinds and numbers of atoms in the smallest representative unit of the substance |
B. Multiple Choice:
11. The correct name for the As-3 ion is the:
a. arsenide ion b. nitrogen ion c. nitride ion d. nitite ion
12. Which element when combined with flourine would most likely form molecular compound?
a. Cesium b. carbon c. potassium d. calcium
13. What is the ionic charge on the oxygen ion in the compound tungsten VII oxide (W2O7)?
a. 2+ b. 2- c. 5- d. 5+
14. Elements of Group 5A and above the stair step:
a. form positive ions. b. form negative ions
c. do not commonly form ions d. do not combine with other elements
15. A row of elements in the periodic table is known as a:
a. period b. list c. group. d. transition.
16. A cation is any atom or group of atoms with:
a. a positive charge
b. no charge
c. a negative charge
d. more electrons that the corresponding atoms.
17. Compounds that are composed two nonmetals:
a. are molecular compounds
b. have relatively high melting and boiling points.
c. are for the most part composed of tow or more metallic elements.
d. fit all of the above descriptions
18. The correct formula for calcium dihydrogen phosphate is:
a. CaH2PO4 b. Ca2H2PO4 c. Ca(H2PO4)2 d. CaHPO4
19. A molecular formula:
a. gives information about molecular structure
b. can be written for ionic compounds
c. shows the number and kinds of atoms in a molecule of a compound
d. shows the number of atoms of each kind by a superscript written after the symbol
20. The nonmetals in Groups 5A, 6A, and 7A:
a. gain electrons when they form ions
b. have a numerical charge that is found by adding the group number to -8
c. all have ions with a 1+ charge
d. lose electrons when they form ions
21. When naming a transition metal that has more than one common ionic charge, the numerical value of the charge is indicated by a:
a. prefix b. suffix c. roman numeral d. superscript
22. In naming a binary molecular compound, the number of atoms of each element present in the molecule is indicated by a:
a. roman numeral b. subscripts.
c. prefixes d. suffixes.
23. The lowest whole-number ratio of ions in an ionic compound is called a(n):
a. molecule b. atom
c. ion d. formula unit
24. An -ite or -ate ending on the name of a compound indicates that the compound:
a. is a binary ionic compound
b. is a binary molecular compound
c. contains a polyatomic anion.
d. contains a cyanide polyatomic ion.
25. What is the formula for hydroselenic acid?
a. H2Si2 b. H2Se c. HSO2 d. H2S
FILL IN THE BLANK: ( BUT NOT ON THE QUESTION SHEET).
26. The ionic charge of sulfur, in the compound Ta2S5 is ___.
27. The ionic charge of an element in Group 2A is __.
28. The group A elements in the periodic table are known as the ___ elements.
29. In a polyatomic ion the -ate ending indicates one more ___ atom than the -ite ending
30. Atoms that have a negative charge are called ___.
31. the name of a monoatomic anion ends in ____.
32. ___ is the systematic name for water.
33. Ternary ionic compounds usually contain one or more ____ ions.
34. Binary molecular compounds are composed of two ____.
35. Acids are compounds that give off ____ ions when dissolved in water.
PROBLEMS:
Name these compounds
36) Ag2Cr2O7
37) CdBr2
38) WO2
39) Zn(ClO3)2
40) CuH2SO4
41) Co(NO3)5
42) LiH
43) AgClO3
44) HgBr
45) FeP
Write formulas for these compounds
(46) diphosphorus pentabromide
(47) carbon monoxide
(48) Uranium IV permanganate
(49) zinc chlorate
(50) calcium hydrogencarbonate
(51) copper (I) hydroxide
(52) ammonium nitrate
(53) dichlorine heptoxide
(54) trisilicon mononitride
(55) ammonium phosphate
(56) lead(II) hydrogen carbonate
(57) Uranium(IV) nitride
(58) selenic acid
(59) Nitrous Acid
(60) Hydrocyanic acid
(61) Cadmium phosphite
(62) Iron(II) oxalate
(63) ammonium silicate
Give the Acid names for the following Compounds
64. H3PO3
65. HBr
66. HClO2
67. HNO2
68. H2SO3
69. H2S
70. H2CrO4
Give the formulas for the following acids
71. Nitric acid
72. Hydrocyanic acid
73. Carbonic acid
74. Permanganic acid
75. Dichromic acid
76. Hypochlorous acid
77. 125.992 Mm/ 3.14 x 108 Mg =____cm/ cg
78. 17.0 km2= ___ in2.
79. 717.5 x 105 in3= ___ km3
80. 111.5 K = ____0F
81. There are 6.02 x 1023 atoms of Gallium in 69.72 grams of Gallium. The density of gallium is 6.00 g/mL. What is the volume, in nm3, occupied by one gallium atom?
82. A quantity of heat equal to 149.55 cal is added to 5.40 g of mercury at 25.0O C. What is the final temperature of the mercury? Specific heat(Hg) = 0.033 cal/g*oC.
83. 25.993 x 10-23 Mg/ns2_________________________________________ pg/year2
84. If 105.000 grams of a silver colored metal are heated to the temperature of boiling water 100.00°C and placed in a styrofoam cup containing 90.000 g of water at 23.3 °C and the temperature of the water - metal mixture reaches a temperature of 27.1 °C. What is the specific heat of the metal?
85. There are four naturally occurring isotopes of the element chromium. The relative abundance of each is: 5124Cr = 5.31%, 5224Cr = 84.76%, 5324Cr = 7.55%, 5524Cr = 2.38%.
Calculate the average atomic mass of chromium.
86. A 199.0 g piece of copper wire is heated and the temperature of the wire changes from 19.0oC to 86.0 oC. The amount of heat absorbed is 686 cal. What is the specific heat of copper?
87. A rock has a mass of 0.25 lb. and displaces 0.015 gal. water. What is the density of the rock in g/cm3 ?
88. The equation for the volume of a cylinder is (3.1415)x(r2) x h, where h is the height of the cylinder and r is the radius. A crane operator needs to move a cylindrical chemical storage tank. The tank has a mass of 3500 Kg. The tank has a diameter of 30 meter and a height of 10 meters the tank is half full of a highly toxic chemical with a density of 5.66 g/mL.
What is the combined mass of the toxic chemical and the tank? If the crane is rated to lift 2325 tons. can the crane lift the tank and the toxic chemical?